|
8/31/2023 0 Comments Carbon electron configuration artHowever, this pattern does not hold for larger atoms. The energy increases as we move up to the 2 s and then 2 p, 3 s, and 3 p orbitals, showing that the increasing n value has more influence on energy than the increasing l value for small atoms. The 1 s orbital at the bottom of the diagram is the orbital with electrons of lowest energy. Figure 6.24 depicts how these two trends in increasing energy relate. In any atom with two or more electrons, the repulsion between the electrons makes energies of subshells with different values of l differ so that the energy of the orbitals increases within a shell in the order s < p < d < f. The energy of atomic orbitals increases as the principal quantum number, n, increases. The specific arrangement of electrons in orbitals of an atom determines many of the chemical properties of that atom. This allows us to determine which orbitals are occupied by electrons in each atom. 
Having introduced the basics of atomic structure and quantum mechanics, we can use our understanding of quantum numbers to determine how atomic orbitals relate to one another. Relate electron configurations to element classifications in the periodic table.Identify and explain exceptions to predicted electron configurations for atoms and ions.Derive the predicted ground-state electron configurations of atoms.At this time,Īre next three lowest configurations and they are written in (low energy -> high energy) order.By the end of this section, you will be able to: Well, if you are not familiar with the very structure of atomic Hamiltonian, we can just concentrate on the energy difference between atomic orbitals and spin-spin interaction. $2s^12p_i^12p_j^12p_k^1$ (since $2s$ and $2p$ have different energy with more than one electron)Īre next three lowest configurations and unfortunately we cannot determine the order of them here. $2s^22p_i^12p_j^1$ (two $p$ electrons have opposite spin) Since 3 $p$ orbitals are equivalent and generally change of interaction between total momentum and atomic environment is smaller than change of kinetic energy/repulsion potential of electrons, by changing states of electrons, If you and your group want to study the structure of atomic Hamiltonian strictly, any other configurations that deviate from Hund's-ground-state will be next excited states, since they produce more powerful momenta-quantum mechanical factors interaction that you cannot figure out yet. You might be confused if you try to understand this using $\sum m$ and $\sum s$ merely, so don't be worry about how to explain the interaction numerically at this time. Here spin-orbit interaction is the one of main ideas of the interaction between total angular momentum and other factors. Atomic Hamiltonian has several terms kinetic energy of electrons + repulsion between electrons + attraction between electrons and protons + (spin-orbit interaction) + (etc). Why? That's the way to lower the interaction between total momentum and any other factors. If we go further, $2s^22p_i^12p_j^1$ where $i,j=x,y,z$ and both spins need to be same. Hund's rule can tell us what the lowest electron configuration is and firstly originated from the way to choose a state of lowest interaction between total momentum and entire atomic environment within given electron configuration.īy Hund's rule, lowest electron configuration of carbon atom is $2s^22p^2$.   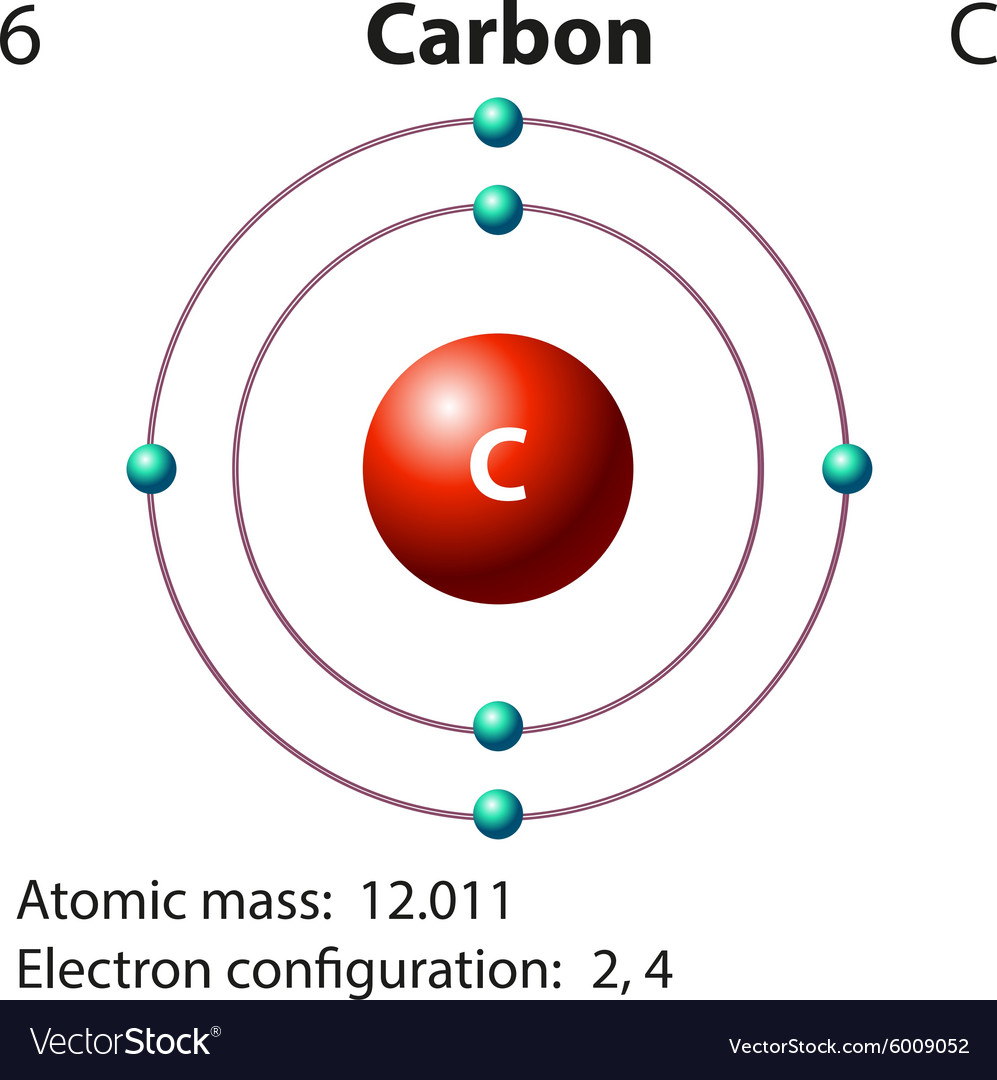
Here you seem to have an idea dealing with angular and spin momenta. The first excited state of carbon $C^*$, and the one that explains the existence of $C(+4)$ chemical compounds, is $2s^12p_x^12p_y^12p_z^1$ where all three lone 2p electrons have the same $m_s$ value.Įlectron configuration means the distribution of electrons in atomic (or molecular) orbitals. $1s^2 2s^2 2p^2$ or with some added detail $1s^22s^22p_x^12p_y^1$ and because $=1s^2$, carbon's electron configuration (ground state) can be written as: Overall we can write the electron configuration of carbon as: Bearing in mind that to satisfy Hund's Rule the latter two 2p electrons are divided over one $p_x$ and one $p_y$ sub-orbital, each with one electron of the same spin quantum number ($m_s=-\frac$). The first 2 occupy the lowest energy atomic orbital possible, that is 1s, so we have $1s^2$ for the first term.įor the remaining four electrons, the next two lowest available atomic orbitals are 2s and 2p and following the above rules that gives us $2s^2$ and $2p^2$. By 'electron configuration' can be understood the way an atom's electrons are arranged in atomic orbitals, in accordance with Pauli's Exclusion Principle, the Aufbau Principle and Hund's Rule, of the lowest possible total energy (known as the Ground State).įor carbon (Z=6), six electron have to be placed in the correct atomic orbitals.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
